 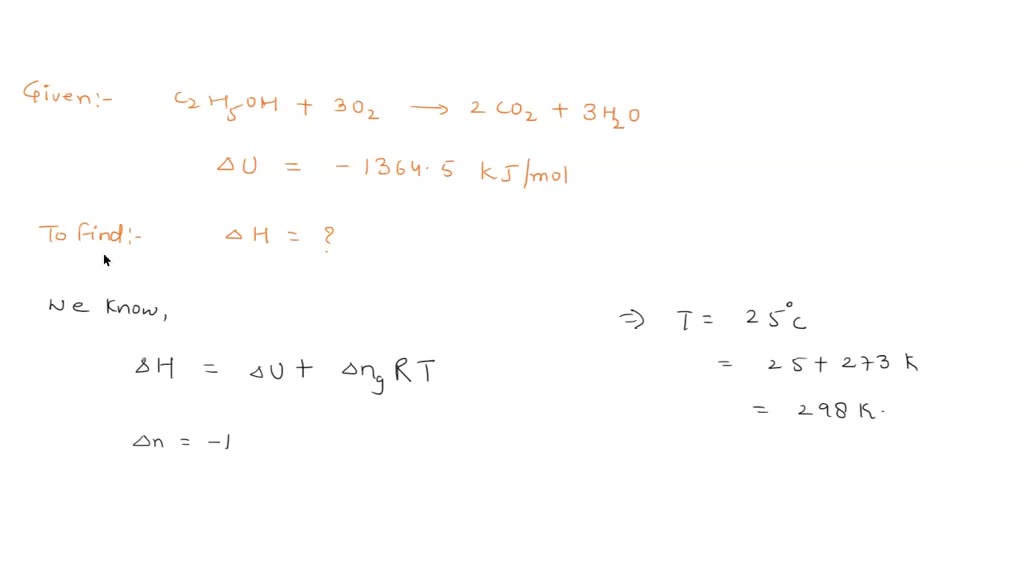
It is obtained from sodium hydrogen carbonate, NaHCO3, by heating. NIST/TRC Web Thermo Tables (WTT) NIST Standard Reference Subscription Database 3 - Professional Edition Version 2-2012-1-Pro This web application provides access to a collection of critically evaluated thermodynamic property data for pure compounds with a primary focus on organics. The Linde Gas Converter is a valuable tool for anyone who works with gases and is looking for a simple and convenient way to convert from gas to liquid. Using the given reaction enthalpy, calculate the heat energy produced by the combustion of one kilogram of methane. Question: Use the following data to calculate the standard enthalpy of formation of solid strontium carbonate SrCO3 (s) SrO (s) + CO2(s) H 234 kJ/mol Hf. Choose a fluid to model: Ammonia, CO2, Dry Air, R134a, R22, R404a, R410a, R507a, Water. Simply choose the gas you want to convert to start the app, then select the calculation units, enter a value and the results appear immediately with no extra clicks or confirmations required. 
In addition, it gives you further information like enthalpy and boiling point and shows how these values vary at different temperatures (0☌, 15☌, 20☌). Linde Gas Converter is a convenient way to convert volumetric to weight-based units for 15 different industrial gases*.Ĭombining usability with a sleek design, this app allows you to easily switch between liquid and gaseous forms. 
Gas Converter Convert from gas to liquid in 3 easy steps Calculate the enthalpy of formation of acetic acid if its anthalpy of combustion to CO2 and H2O is - 867 kJ/mol and the enthalpies of formation of CO2 and H2O are respectively - 393.5 and -285. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
